The Obama administration has removed a major regulatory barrier standing in the way of desperately needed research into medical marijuana.
On Monday, the Office of National Drug Control Policy, which oversees the United States’ overall drug-fighting efforts, announced it will no longer require scientists seeking to study medical marijuana to pass a stifling Public Health Service review process, the Huffington Post’s Ryan Grim reports.
While this may seem like a small step for marijuana research, the recent announcement could pull the lynch pin that opens the floodgates for a wave of future scientific marijuana research that could add to the already growing list of potential medical applications of the substance.
What’s being changed: The review process was an additional hurdle of bureaucracy for non-government-funded researchers to pass before they could legally launch any marijuana-related studies. All studies involving drugs that the Drug Enforcement Administration classifies as Schedule I (no medical value and high potential for abuse) require researchers to obtain pre-approval from the Food and Drug Administration, the DEA and the National Institute on Drug Abuse in order to legally get their hands on the necessary supplies, but the PHS requirement was bafflingly unique to marijuana.
Eliminating this requirement will remove one more time-consuming obstacle standing in the way of medical marijuana research. As the Huffington Post noted, even opponents of reform like Smart Approaches to Marijuana co-founders Dr. Kevin Sabet and former U.S. Rep. Patrick Kennedy have called for this change, although that might have to do with the anti-pot lobby’s desire to increase research into synthetic alternatives to weed.
“The Obama administration has actively supported scientific research on whether marijuana or its components can be safe and effective medicine,” ONDCP spokesman Mario Moreno Zepeda said in a statement, according to Grim. “Eliminating the Public Health Service review should help facilitate additional research to advance our understanding of both the adverse effects and potential therapeutic uses for marijuana or its components.”
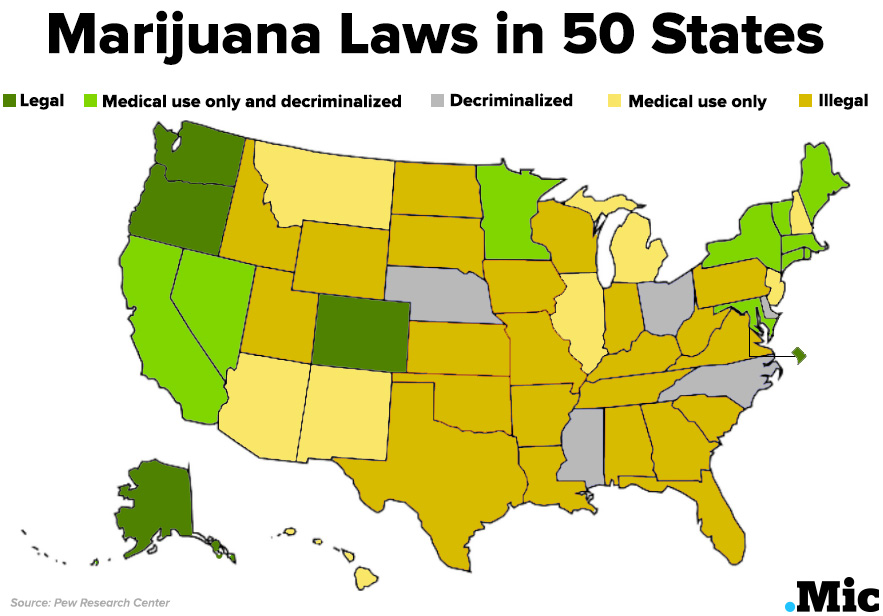
Why it’s important: While state laws on marijuana are rapidly changing, the federal government still had these built-in brakes designed to stall research and other activities that might challenge existing drug control policy.
In addition to passing the now-defunct PHS review, the New York Times reports the three other required pre-approvals are so difficult to obtain that many researchers simply give up. Even when researchers clear all regulatory controls, they are restricted to using a single federally approved source of medical marijuana located at the University of Mississippi — a supply of dubious quality that sometimes simply runs dry.
Removing this barrier to research won’t change things overnight, but it is necessary, given hundreds of studies have found evidence marijuana might be able to aid patients with conditions ranging from brain cancer and Alzheimer’s to PTSD and Crohn’s disease. It’s also a step in the right direction generally, as polling has shown the vast majority of doctors and average people support legal medical marijuana.
Source: Mic.com